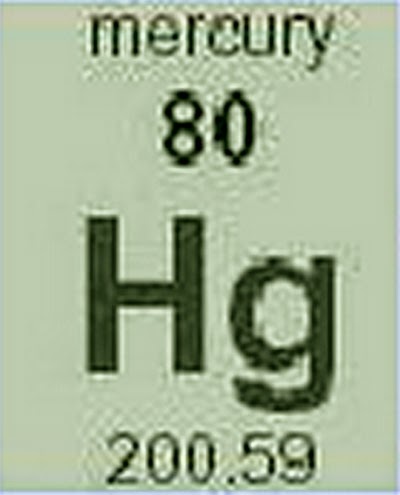
An atom of mercury contains 120 neutrons in its nucleus and 80 electrons around it. Mercury: Symbol: Hg: Atomic Number: 80: Atomic Mass: 200. The atomic number is, while the mass number is. The atomic notation for a particular atom of boron is 11/5B. You will need to refer to a periodic table for proton values. Given the atomic notation of 64/30 Zn, indicate the number of protons, neutrons and electrons respectively, in an atom of zinc. In this notation, the atomic number is not included. Therefore, this quantity has an infinite number of significant digits. 
Recall that the atomic mass of carbon-12 is exactly 12 amu. Symbol-mass format for the above atom would be written as Cr-52. To find the atomic mass of the mercury, multiply 16.716 by the atomic mass of carbon-12. 
For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Alpha radiation travels a very short distance through air, cannot. D) The atomic mass will be between 35 and 37. C) You cannot tell what the atomic mass will be. The atomic mass of chlorine will be more than 37. What is the mass number of an atom of potassium that has 20 neutrons A) 15. Both the atomic number and mass are written to the left of the chemical symbol. Uranium, symbol U, atomic number 92, is in group 6, period P7a of the Periodic Table. the number of electrons in a neutral atom. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
